
Pseudorabies Resources: Latest PRV Fact Sheet (Updated June 2024) & Additional PRV Resources/Links & Statement
As the world deals with the COVID-19 pandemic, SHIC continues to focus efforts on prevention, preparedness, and response to novel and emerging swine disease for the benefit of US swine health. As a conduit of information and research, SHIC encourages sharing of its publications and research. Forward, reprint, and quote SHIC material freely. SHIC is funded by America’s pork producers to fulfill its mission to protect and enhance the health of the US swine herd. For more information, visit https://www.swinehealth.org or contact Dr. Sundberg at [email protected].

Dueling animal health priorities converge as US-based rescue groups work to save dogs from the meat trade in China, bringing them to the US and Canada for re-homing. North American pork industry stakeholders worry about the threat of foreign animal disease transmission posed by these pets, particularly African swine fever (ASF) which continues to be a concern in Asia per Swine Health Information Center Global Swine Disease Monitoring Reports. The supplies used in shipment, such as kennels and bedding materials, are of special concern because of their potential to act as vectors for disease transmission.
The issue of imported dogs from China started to come more to light in mid-2019 through a report from a North Carolina television station. The North Carolina Department of Agriculture State Veterinary office was informed of dogs brought into the state from China. When the Assistant State Veterinarian reached out to the attending veterinarian about the dogs featured on the television report, it was discovered the attending veterinarian was unaware of the significance of the risk these pets represented. State Animal Health Officials worked closely with the attending veterinarian and clinic staff, and subsequent steps were taken to reduce the potential risk of introduction of ASF by in-house quarantine of the dogs in question, disinfection of all materials, bedding, and shipping crates as well as secured disposal and incineration of waste materials.
A February 4, 2021, report by KRTV in Great Falls, Montana, detailed a shipment of 50 dogs flown from China to Los Angeles. After arriving in the US, the dogs were driven to the Canadian border, destined for an unnamed facility. Because there was not a veterinarian available at the Canadian border crossing to check the animals, the dogs were housed at a Great Falls kennel. The report went on to say winter weather extended the stay for a few days. China Dog Rescue has said it plans to bring another 100 dogs into New York state in March 2021 and the organization is also actively raising funds to bring in 60 more soon. And there are also reports of dogs from China arriving in Minnesota, Virginia, and Missouri in addition to North Carolina and Montana.
When considering the importation of dogs into the US for any reason, there are two main federal agencies involved in the importation process. The Center for Disease Control (CDC) and USDA both have responsibilities for the entry of companion animals into the US from foreign countries. Information specific to CDC can be found at https://www.cdc.gov/importation/bringing-an-animal-into-the-united-states/dogs.html. CDC oversees the certificates of veterinary inspection of animals coming to the US as pets whereas the USDA has separate requirements if you are bringing dogs in for the purpose of resale, such as commercial sale or adoption as pets. Information for USDA requirements can be found at https://www.aphis.usda.gov/aphis/pet-travel/bring-pet-into-the-united-states. In either instance, certificates of veterinary inspection are required to accompany any animal entering the US. Additionally, there are possibly more specific requirements depending upon country of origin. Further health requirements for the specific state of destination may also be needed and should be addressed prior to entry to the US.
Veterinarians are the first line of defense against the introduction of FAD and human pathogens with the potential to impact animal health. Veterinary training and knowledge for disease transmission and risk makes the veterinarian the key resource to advise and work with non-veterinary groups toward the protection of animal health. Working closely with all appropriate regulatory agencies and possessing an awareness of international disease threats can prevent unwanted pathogens from negatively impacting clients, farmers and the safety and security of US animal health.
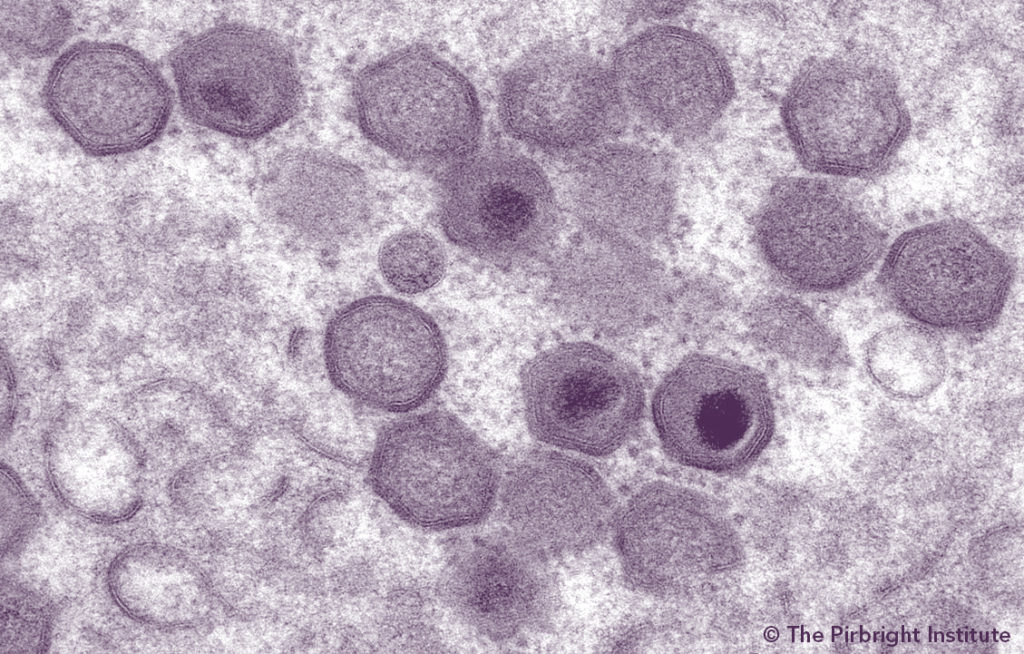
A China Harbin Laboratory paper describing their analysis of African swine fever virus (ASFV) variants emerging in China as well as media and on-the-ground reports of non-approved vaccine use causing pathology, chronic infection and vaccine virus shedding raised concern. The Swine Health Information Center (SHIC) asked Dr. Dan Rock, University of Illinois, to help give some interpretation and context to the Harbin Laboratory paper and attenuating mutations of the ASFV genome such as those that could be used in unapproved vaccines. Dr. Rock has extensive research experience focused on exotic viral diseases of high consequence and the molecular mechanisms that underlie viral virulence.
China ASFV Variants Review by Dr. Dan Rock
In the manuscript “Emergence and prevalence of naturally occurring lower virulent African swine fever viruses (ASFV) in domestic pigs in China 2020.” (Sun et al., 2020), the authors describe genetic changes in all 22 ASFV field isolates collected during a six-month sampling period (June to December 2020) when compared to the virulent field strain, HLJ/18 2020. This manuscript is especially relevant as there are reports of gene deleted vaccine viruses being illegally used in China resulting in chronic infection with increased virus transmissibility.
Notably, 11 of the viruses isolated from two provinces with high pig density contained mutations in the EP402R gene (encoding for the CD2v protein) and exhibited a non-hemadsorbing phenotype (non-HAD). Similar mutations (a single nucleotide substitution resulting in a stop codon and a truncated CD2v protein) were observed in a number of the viruses suggesting they are of the same lineage expressing a truncated CD2v protein of 44 AA. It is likely these represent spontaneous mutations in the CD2v gene occurring on pig-passage rather than engineered mutations in a vaccine virus. Clearly, these mutations are distinct from those previously described for Chinese vaccine virus candidates4; in the case of these engineered viruses, the CD2 gene would be deleted completely with a reporter gene cassette inserted in its place. Given the limited sampling of field virus, the presence of other engineered gene deleted vaccine viruses cannot be formally excluded.
Naturally occurring Non-HAD ASF viruses have been described previously.1, 5, 6, 9, 11 It remains unclear if CD2 mutations occur under selective pressure generated in the host or arise spontaneously and are easily recognized due to loss of the HA phenotype. In any event and consistent with spontaneous mutation, non-HAD ASF viruses appear to arise infrequently. As would be expected, mutations (without easily discernable phenotypes) have been observed throughout the ASFV genome from current European ASFV field isolates separated in time.8
In general, non-HAD ASF viruses including those described by Sun et al.,2020 exhibit altered pathogenesis and attenuation in the pig with the observed effect isolate/infection dose dependent.1-7, 9-11 Typical peracute/acute ASFV infection observed with virulent field isolates is replaced with delayed disease onset and a subclinical and chronic disease presentation characterized by the absence of or reduced mortality and decreased viremia and tissue titers. Duration of viral persistence (blood and tissue) in pigs is variable. The genetic background of the virus may be important for the degree of attenuation/persistence observed for non-HAD ASF viruses as there are examples where CD2v gene mutations/deletions had little to no effect on viral virulence.2, 3, 6, 9
Higher transmissibility was observed for non-HAD viruses evaluated by Sun et al., 2020. Again, this may be isolate/infection dose dependent as the opposite result of reduced transmissibility was observed for Portuguese non-HAD viruses.1 The actual significance of persistent infection with non-HAD viruses and the potential for enhanced transmission under field conditions remains unclear. From available data,1, 10 animals infected with non-HAD viruses seroconvert and are resistant to homologues virulent virus infection.
Non-HAD viruses ASF viruses with reduced virulence and possibly enhanced transmissibility pose new challenges for early ASFV detection and emergency response. Clinical signs observed are not as obvious as those seen with peracute/acute ASFV: therefore, greater vigilance for signs of subclinical and chronic disease is needed. Fortunately, current ASFV diagnostics (serologic and PCR-based) use highly conserved viral structural proteins as targets will detect non-HAD infected animals.
It is important to consider that other attenuating mutations of the ASFV genome such as those that could be used in unapproved vaccines and are yet to be described – not involving CD2v – also may pose problems for early viral detection, response, and disease control of ASF.

During 2019, outbreaks of S. zoo septicemia were reported in Ohio and Tennessee with mortality up to 50%. Then, in January 2021, a sow herd in Indiana experienced abnormally high mortalities related to S. zoo. The emergence of S. zoo associated with mortalities at a level not previously seen in the US prompted the Swine Health Information Center (SHIC) to fund a project conducted at Iowa State University (ISU) and the National Veterinary Services Laboratory to meet the urgent need for information. The result was the first study to experimentally infect and reproduce the disease in weaned pigs with a hypervirulent swine S. zoo strain. Furthermore, pathogenicity differences between genetically different swine strains were described. And a newly developed multiplex PCR provides an accurate and timely assay for detecting and monitoring S. zoo in swine herds.
In the SHIC-funded S. zoo study of the 2019 outbreak, researchers characterized eight isolates associated with high mortality from Ohio and Tennessee by performing whole-genome sequencing on these eight isolates along with another outbreak-unrelated swine isolate from Arizona and 15 S. zoo isolates from other animal species. Phylogenetic analysis showed that the eight 2019 outbreak isolates were clustered together, but genetically distant to the outbreak-unrelated Arizona swine isolate and the isolates from the other animal species. Three full-length complete genome sequences were further assembled, and comparative genomic analyses identified several genomic islands and virulence genes specifically in the outbreak isolates.
The recently completed S. zoo challenge model work evaluated the disease process and diagnostics, which provided meaningful information about the pathogenicity and epidemiology of S. zoo in pigs by challenging one group of conventional six-week-old pigs with hypervirulent S. zoo strains and another group with a genetically different, less virulent S. zoo strain. Additionally, a multiplex PCR assay was designed for S. zoo identification and prediction of virulence in swine isolates.
Then in January 2021, two-year-old adult sows from a production system in Indiana experienced increased death loss. Cyanotic ears, abortion, and uterine discharge was reported and a laboratory diagnosis of S. zoo septicemia was given. With SHIC support provided for further diagnostics these three Indiana isolates were found to be genetically distant and independent to the Ohio and Tennessee isolates, warranting further studies to determine the virulence and to understand the underlying virulence mechanisms.
Findings from these projects provide significant and timely insights for a better understanding of the epidemiology and virulence of S. zoo isolates, consistent with the SHIC mission. In addition, identification of specific virulence genes and genomic islands and the development of molecular diagnostic tool may provide the basis for future investigation of virulence mechanisms and control measures.
Strep equi spp zooepidemicus (S. zoo) was added to the Swine Health Information Center (SHIC) Swine Bacterial Disease Matrix in February 2021 and a fact sheet focused on S. zoo has been added to the SHIC Fact Sheet library. Drs. Raghavendra Amachawadi, Kansas State University, Maria Clavijo, Iowa State University (ISU), Rachel Derscheid, ISU, Connie Gebhart, University of Minnesota, Angela Pillatzki, South Dakota State University, and Nubia Resende-De-Macedo, ISU convened to review the Bacterial Matrix list, score S. zoo for its position on the Matrix and review the new fact sheet. Dr. Kerry Leedom-Larson, ISU, worked with SHIC to do the literature review and drafting for the S. zoo Fact Sheet.
S. zoo received the second highest score of the bacterial pathogens on the Swine Bacterial Disease Matrix. The group considered potential public health impact, the need for more efficacious intervention tools, diagnostic capabilities, potential impact on swine health, welfare and production and potential market impacts as they scored S. zoo for its position on the Matrix.
The Swine Bacterial Disease Matrix goes along with SHIC’s Swine Viral Disease Matrix, a prioritized list of endemic and foreign swine viruses. The Swine Bacterial Disease Matrix offers a focused, prioritized look at the US pork industry’s bacterial risks, consistent with SHIC’s continued focus on emerging pathogens of swine. Both are important tools and part of SHIC’s mission to protect and enhance the health of the US swine herd.
In addition to inclusion on SHIC’s Swine Bacterial Disease Matrix, the new S. zoo Fact Sheet has been developed and posted on the SHIC website. The Fact Sheet models those of the viral pathogens, starting with a summary of important, clinically relevant, information followed by a more complete literature review. It includes information in these categories:
• Etiology
• Importance
• Public Health
• Infection in Swine
• Treatment
• Cleaning and Disinfection
• Prevention and Control
• Transmission
• Pathogenesis
• Diagnosis
• Epidemiology
• Immunity
• History in Swine
• Gaps in Preparedness

Transboundary diseases, including African swine fever (ASF), pose a significant threat to US pork producers. “We know early detection is the key to their control and elimination. But we also know that the approach that served the industry 30 years ago cannot keep pace with today’s big, fast industry, and the 5.2 million pigs that cross state lines each month,” remarked Dr. Jeffrey Zimmerman, Iowa State University. “We need a new surveillance plan – something effective, yet practical and affordable.” Dr. Zimmerman and colleagues at Iowa State University conducted research funded by the Swine Health Information Center (SHIC) and the National Pork Board and found that “spatially balanced sampling” could achieve a higher probability of detection and at lower cost than previous methods.
Dr. Zimmerman says spatially balanced surveillance uses a few samples from many farms across a defined region to determine the region’s status. In contrast, past methods tried to prove that each site was negative to prove a region as negative. “The results have been very promising in terms of developing a better approach for disease surveillance,” he commented. “Work currently in progress by our research team will continue to explore the strength of the approach and its potential to serve the swine industry.”
In their work, five spatially balanced sampling methods were compared to simple random sampling (SRS) in terms of the probability of detection by sample size. Using a livestock disease transmission model in a hypothetical region roughly the size of Iowa and populated with 6000 farms, four of the five spatially balanced sampling methods provided better performance than random sampling. That is, for any given probability of detection, spatially balanced methods required testing fewer farms than SRS.
In an era of pandemics, active regional surveillance for early detection of emerging swine pathogens becomes urgent, yet shrinking budgets impose constraints. To prepare for the introduction of ASF into the US, the USDA Center for Epidemiology and Animal Health in Fort Collins, Colorado, is developing surveillance methods for control areas, for example, determining how best to sample the farm or the population in a barn to verify the farm’s or barn’s disease status. Outside of control areas, spatially balanced surveillance will be critical in verifying freedom from disease and supporting business continuity.
Surveillance efficiency – achieving highest probability of detection at the lowest cost – is central to the public good. This project represents the first step in the investigation of the use of spatially balanced sampling methods in regional livestock disease surveillance programs. The better performance and higher efficiency of spatially balanced sampling methods suggests a real improvement in regional livestock disease surveillance and the challenge of affordable surveillance. Future work will examine the impact of the rate of disease spread and the threshold distance for farm-to-farm transmission on the performance of spatially balanced sampling methods.
As the world deals with the COVID-19 pandemic, SHIC continues to focus efforts on prevention, preparedness, and response to novel
and emerging swine disease for the benefit of US swine health.

This month’s Domestic Swine Disease Monitoring Report shows that porcine reproductive and respiratory syndrome virus (PRRSV) detection was similar to February. A substantial decrease in PRRSV detection in wean-to-market animals was observed. Based on PRRSV detection, ORF-5 sequences, and disease diagnosis reports, and also based on input from the advisory group, the PRRSV 1-4-4 Lineage 1C variant (144 L1Cv) situation is not further spreading or becoming an epidemic. We will continue to monitor the situation closely. March 2021 had a record-high number of cases tested for porcine delta coronavirus (PDCoV) in the US. PDCoV detection was the highest since the PDCoV-epidemic year of 2014. On the other hand, Mycoplasma hyopneumoniae reached the lowest level of detection for the year 2021. At the state level, PRRSV detection was three standard deviations above expected in Nebraska and Ohio. PDCoV was three standard deviations above expected in Kansas, Oklahoma, Missouri, and North Carolina. In a recorded interview, Drs. Trevisan, Magalhaes, and Linhares reflect on the main SDRS findings so far in 2021 and educate the users on how to use the newly launched disease diagnosis web-tool.

In the April Global Swine Disease Monitoring Report, read about a joint USDA/CFIA protocol around the event of African swine fever (ASF) detection in feral swine in Canada or the US. OIE has launched a new World Animal Health Information System interface with enhanced capabilities. In Poland, the first ASF outbreak of 2021 has been reported. An update on the ASF situation in China is shared as well as a report on unlicensed ASF vaccines in Asia.