
Pseudorabies Resources: Latest PRV Fact Sheet (Updated June 2024) & Additional PRV Resources/Links & Statement
The Swine Health Information Center, launched in 2015 with Pork Checkoff funding, protects and enhances the health of the US swine herd by minimizing the impact of emerging disease threats through preparedness, coordinated communications, global disease monitoring, analysis of swine health data, and targeted research investments.
The Swine Health Information Center and the Foundation for Food & Agriculture Research (FFAR) have partnered to fund a $1 million research program to enhance US prevention, preparedness, and response capabilities for Japanese encephalitis virus, a transboundary disease risk for US introduction.
SHIC’s strategic mission to identify risks to swine health includes global and domestic disease monitoring to maintain an acute awareness of emerging swine diseases around the world. Japanese encephalitis is an emerging zoonotic disease identified through global monitoring as a priority for North American prevention and preparedness activities. JEV is transmitted through the bite of infected Culex mosquitoes and biosecurity practices focused on mosquito control are key to reducing risk. The US is currently negative for this mosquito-borne virus which has waterbirds as a natural reservoir but is capable of infecting pigs, humans, and horses.
In 2022, an outbreak of JEV genotype IV spread rapidly across new geographic regions of Australia affecting breeding swine herds and causing reproductive failure, delayed farrowing, stillbirths, mummified fetuses, abortions, and weak piglets. This recent incursion of a new JEV genotype into areas previously free from disease warrants the need for a close investigation of this virus and its potential for incursion and establishment in the US. Understanding the potential impacts of JEV on pork production in the US is critical to protecting the health of the US swine herd as well as mitigating the risk of this emerging disease.
In response to this emerging disease, SHIC and FFAR have partnered to invite proposal submissions from qualified researchers for funding consideration to address identified research priorities for JEV, including topic areas of 1) transmission and epidemiology, 2) mosquito control, 3) diagnostics, 4) communication, 5) surveillance, 6) compatible cases, 7) challenge models, 8) vaccines, 9) cross-protection, 10) competent vectors, 11) role of wildlife, 12) novel hosts, and 13) viral sequencing. Proposal information and a detailed list of research priorities can be found here.
Proposals should clearly state which of the identified SHIC/FFAR JEV research priorities will be addressed through the project. Collaborative projects that include the pork industry, international organizations, allied industry, academic institutions, and/or public/private partnerships are highly encouraged. Projects demonstrating the most urgent, timely completion, providing the greatest value to pork producers, and showing efficient use of funds will be prioritized for funding. Projects are requested to be completed within a 12-to-18-month period with sufficient justification required for extended project duration. The JEV research proposal template can be found here.
Total funding available for the SHIC/FFAR JEV Research Priorities is $1 million. Individual awards are capped at $250,000 but proposals may exceed the cap if sufficient justification is provided. Matching funds are encouraged but not required; the $250,000 cap applies to only those funds requested from SHIC/FFAR. All projects should strive to be unique, have a high impact, show value to pork producers, and have industry-wide benefit.
The deadline for proposal submission is 5:00 pm CDT on April 15, 2024. SHIC and FFAR are co-hosting an informational webinar on February 22, 2024, at 3:00 pm CST to provide additional details about the application criteria. Additional information can be found at www.swinehealth.org. For questions, please contact Dr. Megan Niederwerder at [email protected] or (785)452-8270 or Dr. Lisa Becton at [email protected] or (515)724-9491.
The Foundation for Food & Agriculture Research (FFAR) is a non-profit organization established in the 2014 Farm Bill to build public-private partnerships that fund bold research addressing food and agriculture challenges. SHIC and FFAR collaborated with the Pork Checkoff to fund the Wean-to-Harvest Biosecurity Program research project now underway.

The Swine Health Information Center and the Foundation for Food & Agriculture Research (FFAR) partnered to fund a $1 million research program to enhance US prevention, preparedness, and response capabilities for Japanese encephalitis virus (JEV), a transboundary disease risk for US introduction. SHIC and FFAR co-hosted an informational webinar on February 22, 2024, regarding the request for proposals for the program. The webinar recording can be viewed on-demand. Deadline for proposal submission is 5:00 pm CDT on April 15, 2024. Awards for this program are anticipated in late spring 2024.
FFAR staff participating in the webinar included Lauren Hershey, director of strategic partnerships, Dr. Jasmine Bruno, scientific program director and leader of the animal systems research portfolio, and Michelle Olgers, communications officer. They were joined by SHIC Executive Director Dr. Megan Niederwerder and SHIC Associate Director Dr. Lisa Becton. The goal of the joint research program is to develop novel tools and technologies enabling the US to prevent introduction of JEV. Additionally, preparation for a potential JEV incursion will include development of a response strategy and identifying ways to mitigate production losses should JEV be introduced in the US.
With significant solicitation of ideas and stakeholder feedback, SHIC and other partners developed 13 research priorities for the JEV program. Priorities include transmission and epidemiology of the virus, developing mosquito control strategies for US hog farms, development and evaluation of diagnostic tests including genome-based and antibody-based options, developing effective communication strategies for producers and consumers, surveillance plans and identifying surveillance targets, evaluating compatible cases that would be submitted to veterinary diagnostic labs in the US, developing challenge models to investigate interventions and their impacts on disease, identifying novel vaccine strategies, and understanding the potential for cross protection from other US flaviviruses. Other priorities include identification of competent vectors in the US, understanding the potential role of wildlife in JEV transmission and spread, defining novel hosts that could support viral replication, and evaluating viral sequencing to understand the molecular pathogenesis of JEV.
The mechanics and content requirements for proposal submission were also covered during the webinar. Dr. Becton noted the SHIC proposal template is different from the FFAR template and contains unique requirements for submission under this program. Specifically, proposals should address one or more of the listed research priorities. Project duration should be 12 to 18 months. However, if there is a compelling need for additional project duration, justification for the extended time should be included within the body of the proposal.
The maximum award per submitted proposal is $250,000. SHIC and FFAR encourage matching funds from other entities, but it is not required. Matching funds can be noted in a separate column on the budget template. SHIC and FFAR encourage proposal submissions that include collaborations across industries, including international organizations, allied pork industry partners, academic institutions, and other private and public partnerships. All proposals must provide research outcomes that benefit US pork producers and the US swine industry.
The proposal template requires the proposal body to be five total pages, typed in single-spaced, 12 point Times New Roman font. Proposal introductions should include project rationale, the specific priority being addressed, and a brief listing of project objectives. As the second part of the proposal, the research design should clearly and comprehensively state the details of the experimental methodology to accomplish each objective. Details should include reference assays or tests being utilized, sample types including frequency of collection, and statistical tests that will be employed for data analysis. This section is critical for reviewers to understand the research plan and evaluate scientific merit. Objectives should provide a clear format for data generation and be able to answer the key hypotheses within the proposal.
A description of the quality assurance and quality control plans should be incorporated into the proposal, including the steps investigators will take to ensure the project is completed in a standardized, consistent, repeatable, and accurate format. This may include a description of how testing is standardized, how staff or personnel will be trained, and other related steps. Investigators should detail the operating timeline lined out by key activities and milestones. For example, some investigators utilize a Gantt chart to signify key milestones by month. All proposals should include contact information for key persons managing research contracts on the cover page. This information is critical to connect SHIC/FFAR with the appropriate contacts for contract processes once awards have been granted.
On the budget template, specific sections are provided to detail individual costs that may occur for the project, including travel and publication. Overhead or indirect costs are typically not covered by SHIC research funds. SHIC funding can include graduate student support, student hourly labor, and other post-doctoral support. Regarding principal investigators, if appointments are less than 12 months, funding requests must commiserate with the time being spent on the project. SHIC typically does not cover equipment unless it’s approved in advance. In these cases, justification should be included within the budget section for equipment expenses.
Letters of cooperation from stakeholders, co-investigators, and/or institutions who have agreed to work on the project can be added within the proposal but are not counted as part of the five-page limit. The cover page and budget page are in addition to the five-page proposal body. Investigators are also welcome to include a two-to-three-page biosketch and/or references at the end of the proposal. All proposals should be submitted as a single word document.
SHIC highlighted some considerations for proposal review, including the value of research to the US pork industry, addressing one or more of the 13 research priorities, and project relevance and impact to US pork production. Experimental design review can include an evaluation for clearly outlined aims and objectives, inclusion of appropriate, trackable, timely, and feasible methodology, and the presence of a qualified research team. Reviewers will evaluate if the objectives are achievable and if the budget requests are justified. Multiple proposals from the same principal investigator/co-principal investigator, and consortium applications are welcomed. US-based collaborator(s) are encouraged but not required. If projects have international lead investigators, the proposal should clearly demonstrate that the research provides value back to US pork producers and the US swine industry. For additional questions or to review the template and RFP, please refer to https://www.swinehealth.org/call-for-research/ for the SHIC/FFAR JEV RFP.
The Foundation for Food & Agriculture Research (FFAR) builds public-private partnerships to fund bold research addressing big food and agriculture challenges. FFAR was established in the 2014 Farm Bill to increase public agriculture research investments, fill knowledge gaps and complement the U.S. Department of Agriculture’s research agenda. FFAR’s model matches federal funding from Congress with private funding, delivering a powerful return on taxpayer investment. Through collaboration and partnerships, FFAR advances actionable science benefiting farmers, consumers and the environment.

In early 2022, Japanese encephalitis virus (JEV) expanded into new regions of Australia, affecting over 80 swine breeding farms and causing significant production losses. Having never been detected in US swine, JEV poses an emerging transboundary threat to domestic sow herds susceptible to viral infection. As part of industry preparedness activities for JEV, SHIC funded an economic assessment to explore the potential impacts for introduction and establishment of JEV in the US. Dr. Liz Wagstrom, Wagstrom Consulting, led the investigation, alongside a team of economists and production veterinarians, to gather production data during the Australian outbreak and develop a prediction for production losses due to reproductive outcomes during a potential US incursion.
Now complete, the full domestic JEV incursion economic assessment white paper can be accessed here. Authors estimated that 32% of the US sow herd would be at-risk for JEV infection and that resulting sow herd losses would range from 1-2% of production. Assuming no increase in prices due to the diminished output, economic losses to the US pork industry would be between $306 million and $612 million. While the study did not assess the impact on boar studs or changes in demand for pork and pork products, the predicted production impact for US swine herds supports the need for continued JEV prevention and preparedness activities.
The 2022 outbreak of JEV genotype IV spread rapidly across new geographic regions in Australia affecting breeding swine herds and causing reproductive failure, delayed farrowing, stillbirths, mummified fetuses, abortions, and weak shaker piglets. Additional clinical signs were noted for infected boar studs including orchitis, decreased sperm number (in some cases progressing to semen with no sperm) or motility in semen, and abnormal spermatozoa. Between February 25 and March 3, 2022, JEV infection in pigs was confirmed in four Australian states. By mid-2022, more than 80 pig farms located over much of the southern and eastern parts of Australia were infected. Since the last identified JEV infection in Australian swine was in a single gilt in November 2022, no additional JEV infections in Australian domestic swine have been identified during 2023 – 2024. Through interviews with Australian veterinarians and analysis of production data, it was estimated that approximately 60% of Australia’s sow herds were impacted with overall production losses ranging from 3% – 6%.
As part of the economic assessment for a potential JEV incursion to the US, the investigative team evaluated differences between the US and Australian pork industries. Compared to the US inventory of 72.2 million swine on over 60,000 farms, the Australian pig industry is smaller with an inventory of 2.4 million swine on 4,300 piggeries. Further, farm ventilation type was compared as it had been previously hypothesized as a risk factor for infection during the Australian outbreak. Australian veterinarians estimated that of the 90% of sows raised in indoor confinement, only 10% are housed in mechanically ventilated facilities. This contrasts with US sow herds predominantly raised in mechanically ventilated facilities including filtered barns, which are believed to have lowered risk for mosquito exposure. For US herds, ventilation type varies by production region and was considered when assessing overall percentage of sows impacted by a potential JEV incursion.
The total estimated number of sows predicted to be impacted in the US was 2,135,940, which is equal to 32% of the total US breeding herd. As affected farms in Australia experienced a 3% – 6% reduction in annual output, applying the same 3% – 6% value across the number of impacted US sows suggests a reduction in overall US production output of 1% – 2%. Cash receipts for the US pork industry totaled $30.6 billion in 2022. This suggests that the economic losses in impacted herds would be between $306 million – 612 million in a 1-year outbreak scenario. For a 2,500 head sow farm that had achieved 30 weaned pigs per sow per year prior to the outbreak and assuming each weaned pig is worth $40, the production losses amount to $90,000 – $180,000 per year.
Understanding the potential economic impacts of JEV on pork production in the US is critical to protecting the health of the US swine herd as well as mitigating the risk of this emerging disease. While certain production losses were not assessed, including impact on trade or boar stud output, the information gained in the report illustrates the potential negative impact for US pork production on sow farms. This economic estimate supports the need for research to understand the dynamics of disease transmission and the development of strategies for prevention and control. Critical research investments continue to be necessary to prevent JEV incursion, ensure rapid detection of JEV if introduced, inform stakeholder response, mitigate production losses on sow farms, identify effective control measures, and develop clear messaging to consumers on the safety of pork.
In January 2024, the Swine Health Information Center issued a formal request for proposals inviting submissions to specifically address 11 of the 36 research priorities and topics published in its 2024 Plan of Work. Areas of research spanned across three of the Center’s five strategic priorities, including monitoring and mitigating risks to swine health, responding to emerging disease, and surveillance and discovery of emerging disease. Proposals were due March 1, 2024, and a competitive review process for value to the pork industry by a SHIC working group will provide funding recommendations. A total of 43 proposals from 21 institutions were received by the deadline.
“Through targeted research investments, SHIC is striving to drive innovation in the pork industry for the protection and enhancement of US swine herd health,” remarked SHIC Executive Director Dr. Megan Niederwerder. “The formal RFP received an outstanding response, providing an opportunity to recruit a broad net of research ideas to generate new knowledge in the field of emerging swine diseases.”
Research outcomes from the projects to be funded from this request for proposals will provide critical information and resources to help pork producers as they face emerging disease challenges in their swine herds. Research priorities and topics identified in the SHIC 2024 Plan of Work help the organization fulfill its mission to generate new intelligence for preventing, preparing, and responding to emerging swine disease threats. Funding available for the SHIC 2024 Plan of Work Research Program priorities included in this RFP is $1.1 million. Individual awards are anticipated to be between $50,000 and $150,000.
The specific research priorities included in the January RFP were high-risk product importation and traveler entry at US borders, cull sow and secondary market biosecurity, engineering biosecurity through site construction or renovation, disease spillover risks from wean-to-market pigs to sow herds, understanding the impact of porcine circoviruses, identifying early disease outbreak warning signals, diagnostic assays for confirming cleaning/disinfection protocols, expansion of population based sample types for emerging disease testing, pan-diagnostic assay development, environmental sample types for emerging disease surveillance, and investigating the clinical relevance of newly identified agents from veterinary diagnostic lab submissions.
Through the RFP process, SHIC’s goals were to encourage researchers to develop and submit proposals that specifically addressed the identified priorities, broaden awareness of funding opportunities to advance its 2024 Plan of Work, and expand the scientific network of researchers and institutions conducting critical research on emerging swine diseases. Funding timely research is an essential component of SHIC providing project outcomes that drive action for emerging disease prevention, preparedness, and mitigation.

On February 6, 2024, the Swine Health Information Center along with the American Association of Swine Veterinarians offered a webinar focused on emerging trends of porcine circovirus. Expert presenters provided the latest information on porcine circovirus including domestic and global distribution, new research updates, diagnostic trends, sample types submitted for surveillance, interpretation of test results, and practitioner perspectives for disease management strategies.
There were 311 people registered for the PCV webinar, conducted by the Swine Medicine Education Center at Iowa State University, which is now available online for review. The first presenter, Dr. Tanja Opriessnig, ISU, provided a general disease overview including domestic and global distribution, recent research outcomes, and an update on PCV4. She said although PCVs were first identified approximately 27 years ago, an improved understanding of circoviruses and their impact on swine health has continued to evolve over time.
PCV2 has been a significant health challenge for global pig producers for many decades. Dr. Opriessnig said PCV2-specific vaccines have worked remarkably well to prevent viral health impacts since global introduction in 2006. Modern PCV2 vaccines provide cross-protection against currently known PCV2 genotypes and are among the most widely used vaccine in growing pigs. Dr. Opriessnig shared evidence of the global distribution of PCV3 as well as the recent identification of PCV4 in Asia and Spain, saying a more field-based approach is needed for prevention, preparedness, and response to emerging PCVs. Dr. Opriessnig remarked that initial evidence shows that PCV3 vaccines are helpful for control and that monitoring for emerging strains, including porcine circovirus-like viruses, is ongoing.
Dr. Darin Madson, ISU Veterinary Diagnostic Lab, shared background information for PCV2, stating it is a globally distributed and ubiquitous virus with very few negative swine herds in the US. PCV2 is an economically important disease, ranking as one of the top three infectious agents in the swine industry. Dr. Madson remarked that infection generally occurs after waning of maternal antibodies in pigs 7- to 15-weeks of age. Transmission of PCV2 is possible both horizontally (pig to pig) and vertically (dam to offspring) through nasal-oral and fecal-oral routes. He pointed out a major route of transmission occurs through virus secreted and/or excreted in urine, ocular discharge, nasal discharge, saliva, and feces.
Fomites capable of transmission include needles, insects, and rodents. Aerosol transmission of PCV2 is possible as the virus has been detected in exhaust air and dust outside barns; however, infectivity is unknown. The virus is environmentally hardy and difficult to inactivate with disinfectants.
During his presentation, Dr. Madson shared an AASV Board-approved porcine circovirus position statement to aid in accurate diagnosis of PCV.
PCVAD can be subclinical or include one or more of the following clinical manifestations concurrently:
For diagnosis, the following signs are triggers:
Dr. Madson discussed the continued challenge of determining PCV2 infection vs disease. He stated the diagnosticians’ dilemma is interpreting positive test results without clinical disease or lesions. His presentation also included a review of PCV2 immunity and reviewed information about possible causes of disease control lapses and immunity failures.
Dr. Pablo Pineyro, ISU VDL, provided further information regarding PCV2 vaccination and impact on reducing viral load in serum and tissues, albeit not completely stopping PCV2 antigen expression. Dr. Pineyro shared knowledge gained from VDL submissions, stating the amount of PCV2 DNA and antigen detected by PCR and IHC strongly correlates with vaccination status. Genetic changes of the virus over time can affect vaccine protection and vaccination challenges should be evaluated in association with genetic changes and biological relevance on the farm. While PCV2 vaccine has reduced PCVAD clinical prevalence, it has not changed the challenges associated with PCVAD diagnosis. For PCVAD diagnosis, a correct interpretation of the result depends on the clinical question and the diagnostic tool.
When considering PCV3, the challenge is to determine clinical relevance, including causation vs detection. Currently, there is a lack of specific lesions and direct detection tools for PCV3. Clinical cases associated with infection and multi-systemic inflammation (grower-finishers) are all being considered, Dr. Pineyro noted.
Dr. Mark Ladd, Smithfield, shared his experience with PCV detection, treatment, and management from 2014-2023. During this time, he oversaw 65,000 sows and all related downstream nurseries and grow/finish units. In 2014, he started using sequencing to identify PCV2 genotype and evaluate vaccination status. If PCV2b was detected, Dr. Ladd concluded either animals were not vaccinated, if seen in multiple ages with a specific flow, or there was a vaccine failure which was usually an individual farm problem. If PCV2d was detected, Dr. Ladd would not look at vaccination status as the primary cause. Calling it the PCV2d puzzle, Dr. Ladd noted vaccinated animals can still be viremic but may or may not have clinical disease as the virus can be found in healthy pigs. These characteristics make diagnosing reproductive failure by PCV2d very difficult, per Dr. Ladd.
Sharing his experience with growing pigs, Dr. Ladd said elevated mortality and a lack of response to antibiotics were factors initiating tissue collection for a diagnostic workup. Suspect PCV cases with either lesions consistent with PCVAD or PCR Ct values less than 25 raised concern. Dr. Ladd said 6% of the PCV2 cases he saw were from nursery pigs (3- to 10-weeks of age) and 94% of the cases were from finishing pigs (11- to 31-weeks of age). Among the PCV2 cases, 88% were also PRRSV positive and 25% were IAV positive, with only 5% having neither PRRSV nor IAV. Dr. Ladd said flows returned to normal when PRRS and/or IAV were addressed. Methods to stabilize the herds included depop/repop if it was a continuous flow growing site or stopping PRRSV circulation upstream at sow farms. IAV vaccine was used after determining strains found in the herd. His experience leads Dr. Ladd to say it appears PCV2d needs co-infection to be clinically relevant. And he asks, does co-infection with PRRS and/or IAV increase the possibility of PCV2 vaccine failure in pigs?
Dr. Chelsea Stewart, Christensen Farms, began by asking if the detection of PCV2 is clinical, subclinical or irrelevant? In her experience, PCV2 was occasionally detected in certain flows without PCVAD clinical signs. She noted significant stressors such as PRRSV, poor ventilation, weather changes, and delayed vaccination appear to be risk factors. When PCVAD was present, signs included CT values in the low teens, lymphoid depletion, wasting, and fallout.
In response to PCVAD in one commingled flow, Dr. Stewart initiated field exposure surveillance in cooperation with ISU VDL and Boehringer Ingelheim. A cross-sectional surveillance of downstream flows was to determine the timing of exposure and if the existing vaccination timing made sense. Sow farm surveillance included collecting processing fluids in one room per week for 12 weeks. Dr. Stewart said this was designed for long-term, system-wide surveillance. From the processing fluids, the VDL sequenced several positives with most being PCV2a or PCV2d, with a few PCV2e. As a result, Dr. Stewart’s overall question was, can we correlate PF PCR CT values to sow immune status to gauge efficacy of the current vaccine protocol?
Another step in their process was to determine individual processing fluid correlation to sow immune status. To measure this, two sow farms with PCR positive results and a high rate of fetal mummification were chosen for further investigation. Processing fluids were collected from litters and serum from sows. Sows were equally divided with 10 at parity one, 10 in parities two through five, and 10 at parity five or more. Dr. Stewart said no cross fostering was allowed until after processing fluids were collected. Processing fluids were evaluated for PCV2 by PCR and serum was used for PCV2 antibody testing. Goals of the study were to evaluate if CT value correlated to individual sow status or parity.
Dr. Stewart concluded by saying they did not detect a parity difference but did identify a gap in gilt immunity that may have resulted in the PCVAD cases. As a result, they modified their vaccination program to include booster vaccines at the sow farms and a rescue dose to growing pigs between 5- and 9-weeks of age. Flow has continued to perform around target since discontinuing the rescue dose and sows are a full gestation past whole herd booster. Vaccination compliance is critical, per Dr. Stewart who also recommends not chasing positive CT results without a clinical diagnosis.
PCV presents a significant swine disease challenge and is included in SHIC’s 2024 Plan of Work as a priority pathogen.

The Swine Health Information Center continues to share information on its strategic priorities and mission to protect the health of the US swine herd with veterinarians and pork producers. During the recent American Association of Swine Veterinarians Annual Meeting, February 24-27 in Nashville, Tennessee, over 1000 attendees had the opportunity to learn about SHIC’s work through session presentations, committee participation, and a tech table display.
As part of the pre-conference committee meetings, SHIC Executive Director Dr. Megan Niederwerder and SHIC Associate Director Dr. Lisa Becton presented SHIC’s 2024 Plan of Work and Requests for Proposals on research priorities to the Committee on Transboundary and Emerging Diseases. Both Drs. Niederwerder and Becton attended the pre-conference session on biosecurity and participated in the SDRS advisory group meeting where new information about this SHIC-funded disease monitoring report was shared.
During AASV’s special session, “Beagles and Bagels,” Dr. Niederwerder presented the latest information on Japanese encephalitis virus, including the current JEV status in Australia, the availability of JEV testing at National Veterinary Service Laboratories, progress on the updated USDA response strategy, the SHIC/FFAR JEV Research Program, and updates on the risk and potential impact that JEV poses to the US pork industry. She also updated attendees about SHIC research activities on emerging diseases and collaborative partnerships with the other pork industry organizations at the AASV annual business meeting.
The results of SHIC-funded research and programs were featured in several seminars, on research posters, and during presentations, including in the general session. A tech table display featured SHIC projects and tools for communicating the research results to help keep the US swine herd safe and stay ahead of economically devastating health issues. Interactions with AASV participants were instrumental in furthering SHIC’s outreach and expanding the network for SHIC as a resource to help herd veterinarians, pork producers, and researchers work on emerging swine diseases.

This month’s Domestic Swine Disease Monitoring Report includes the new SDRS Blast for PRRSV ORF5 sequence tool. The newly implemented tool aids veterinarians and producers in identifying their own PRRSV ORF5 sequences’ similarity with previous PRRSV sequences in the SDRS database. Also, the report brings information about the first PRRSV L1C.5 detection in South Carolina. The detection was in a sample identified as having been collected from a finishing site, with 100% nucleotide identity with another PRRSV ORF5 sequence recovered in another state and identified as L1C.5 RFLP 1-4-3. For enteric coronaviruses, PEDV positivity had a substantial increase during February in the wean-to-market category. PDCoV positivity remains high in the wean-to-market category, similar to the increased positivity of January 2024. Mycoplasma hyopneumoniae substantially increased positivity for the adult/sow farm category. However, according to the SDRS advisory board, this activity is due to few farms and does not represent abnormal activity in sow farms. In the podcast, the SDRS hosts talk with Dr. Lauren Glowzenski (JBS) about solutions for decreasing PEDV activity in farms and PRRSV ORF5 sequence regional diversity.

In this month’s Global Swine Disease Monitoring Report, read about Albania which has become the 28th European country to confirm the presence of African swine fever, genotype 2, since the virus was introduced to the continent in 2007. Details on wild boar population control measures in Europe are included. Authorities in Italy, Poland, Estonia, and Norway have issued statements outlining measures to control the wild boar population as a strategy to combat ASF. Learn about nearly 10% of Chinese meat products testing positive for ASF during Taiwan’s border inspection. Strict border inspections have been effective in preventing high-risk products from entering, as evidenced by a decrease in fines from 3.61 cases per 10,000 passengers to 2.85 from 2019 to 2023, respectively. The report includes information on WOAH’s thematic study on zoning. WOAH has launched its first thematic study through the Observatory program, focusing on the international standard of zoning. The study assesses the implementation of zoning for avian influenza, ASF, and foot-and-mouth disease, highlighting its use, challenges, and impacts.
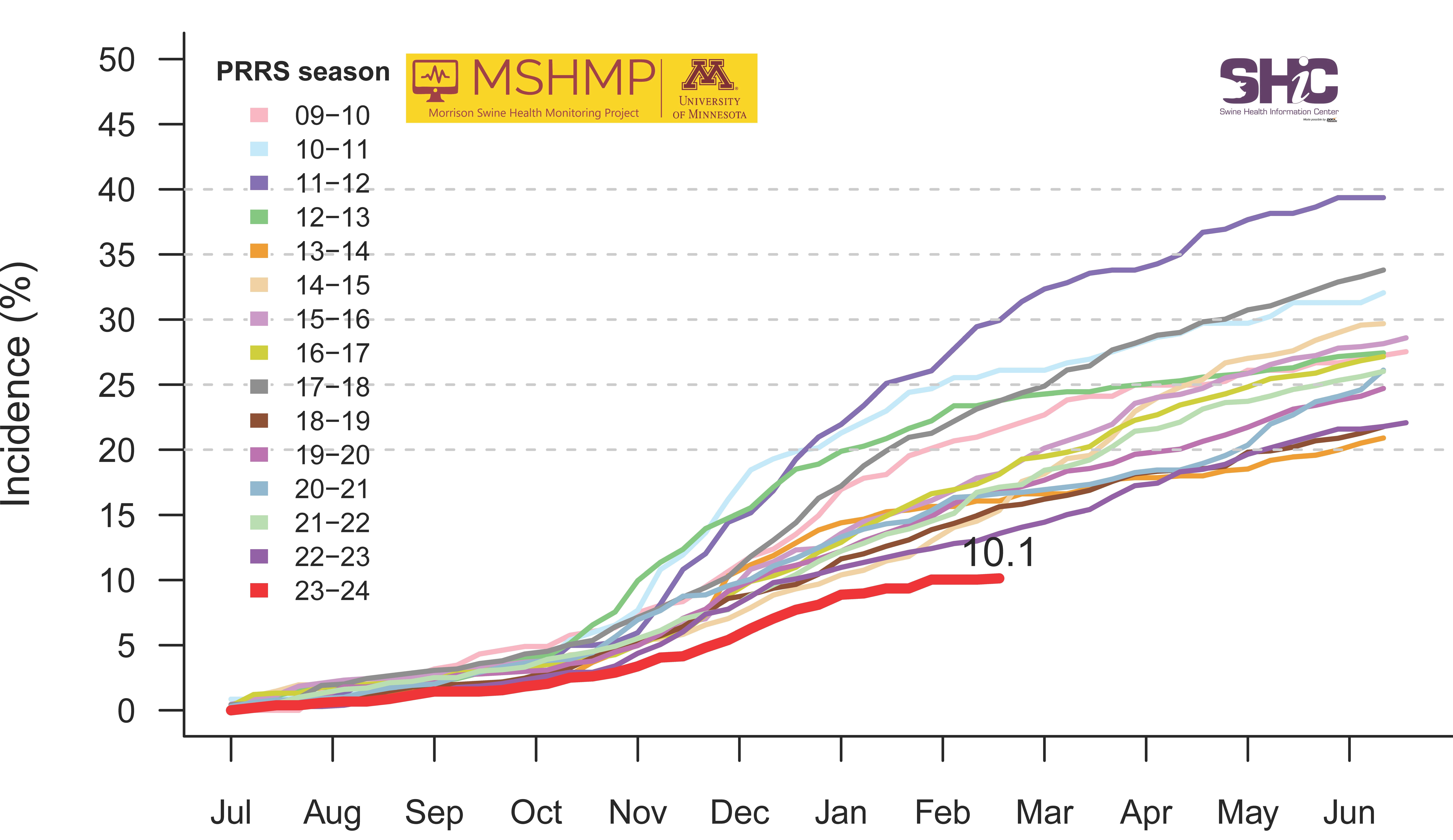
PRRS Cumulative Incidence for MSHMP Beginning July 1, 2009
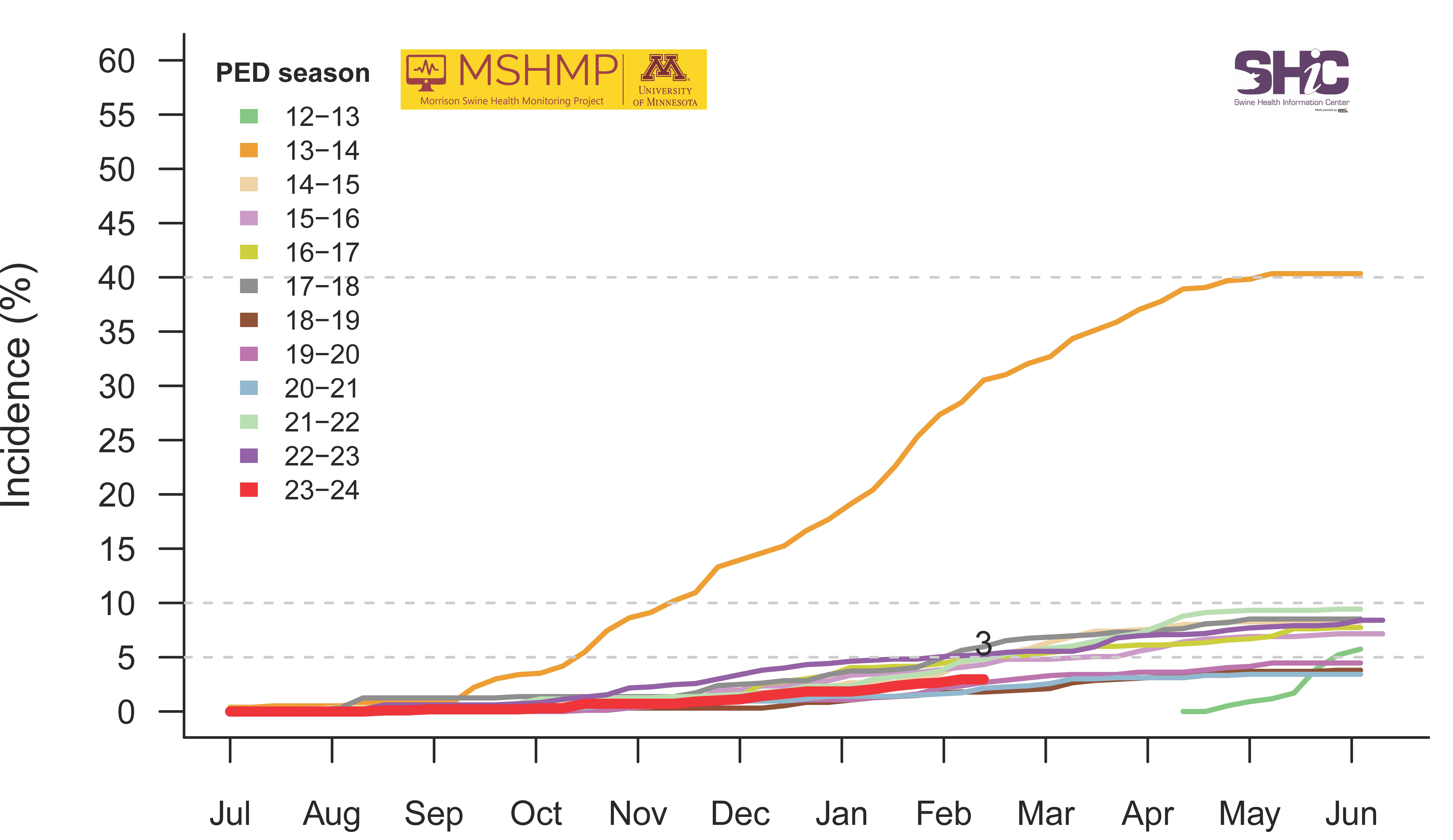
PEDV Cumulative Incidence for MSHMP Beginning May 1, 2013