
The Swine Health Information Center, launched in 2015 with Pork Checkoff funding, protects and enhances the health of the US swine herd by minimizing the impact of emerging disease threats through preparedness, coordinated communications, global disease monitoring, analysis of swine health data, and targeted research investments.

Click here to learn more about SHIC’s project to better understand and reduce the risk of ASF spreading through feed ingredients as highlighted by federal partner USDA NIFA.

The first Global Disease Monitoring Report (GDMR) was published in November 2017 to provide US pork producers with a clear overview of the global situation of transboundary swine diseases and support informed decision-making. At the time, access to reliable and timely information was a major challenge as many outbreak reports were scattered across local-language sources, difficult to locate, time-consuming to verify, and often delayed or limited in detail in official reporting systems such as WOAH. The report aimed to synthesize and verify this information, bringing key developments together in one accessible place, made possible by Swine Health Information Center support and funding in collaboration with the University of Minnesota.
Read the special 100th Global Swine Disease Monitoring Report review here!

Japanese encephalitis virus, a mosquito-borne virus, can infect many species including swine and humans. While JEV is not currently in the US, incursion risk remains a concern given the virus’s recent global expansion. The presence of JEV in the US could have devastating outcomes for the swine industry because of its severe disease presentation in neonatal piglets and pregnant sows. In the event of an outbreak, accurate JEV diagnostics will be critical to disease monitoring. Currently, no approved diagnostic tests for JEV antibodies in swine exist in the US. As part of the Swine Health Information Center JEV Research Program, funded in collaboration with the Foundation for Food & Agriculture Research, a recently completed project led by Dr. Katharine Bossart, Integrated Research Associates, LLC, sought to build diagnostic testing capabilities for JEV in the US.
The main goal of the project was to build diagnostic capability for JEV based on diagnostic serological assays used and validated throughout Southeast Asia. JEV is classified as a biosafety level 3 agent, which limits the number of laboratories able to work with the virus and further reduces JEV diagnostic capability. Recognizing this, Dr. Bossart and team evaluated and developed JEV serology assays based on similar tests utilized overseas. These assays use nonhazardous recombinant JEV virus-like particles as viral antigens to broaden access and increase JEV testing capacity.
Read the industry summary for this study, #24-061, here.
JEV has been found throughout Asia and the Western Pacific with many of these countries having active surveillance programs in place for mosquitoes and swine. In countries outside of the region, including the US, JEV surveillance programs have not been implemented and vaccination is only advised in those traveling to countries where JEV is endemic. In 2022, a JEV genotype IV virus caused a widespread and devastating outbreak in eastern Australia in commercial piggeries and in humans. Currently, there are no approved diagnostics for JEV antibodies in humans or animals in the US. Initially, recombinant JEV virus-like particles (VLPs) were being made using a novel technology developed to explore their potential as JEV vaccine candidates. Expanding the use of recombinant JEV VLPs to replace inactivated JEV in diagnostic kits was a logical step towards not just building JEV diagnostic capability but also simplifying kit manufacturing.
To complete this study, sufficient quantities of inactivated JEV viral antigens, JE1, FE1, JN1, JN2 and FN1 monoclonal antibodies (mAbs) and corresponding mAb hybridomas, were imported from Venture Technologies. Stable cell lines expressing JEV VLPs from genotypes III, IV and V were used to produce recombinant JEV VLPs. Although recombinant JEV VLPs were made from genotypes III, IV and V, there were unique properties for each that were most notable for JEV V. Results indicated that JEV VLPs performed well and can replace inactivated JEV in diagnostic kits. Multiple genotypes of JEV can be included as antigens, which represents an improvement in design, and a scalable cGMP-compliant manufacturing process has been developed for recombinant JEV VLPs, which will simplify future regulatory filings and approvals.
Two diagnostic prototype kits containing the new JEV antigens were assembled as part of this trial. One is an ELISA kit for screening numerous serum samples in the laboratory and the second is a dot enzyme immunoassay kit for screening individual, or a few, serum samples on farm sites without advanced technology. Prototype kits are currently being evaluated overseas using approximately 500 swine sera from clinically confirmed cases of JEV and results should be available in late summer 2026.
If a large-scale JEV outbreak occurs in the US, the possibility to utilize vaccination exists. In this scenario, the ability to differentiate vaccinated and infected swine becomes important to biosecurity and control. Therefore, the last objective of the project focused on determining if a recombinant JEV nonstructural protein could be used as an alternate antigen in diagnostic kits as only infected people or infected animals (not vaccinated) make an immune response to nonstructural proteins. A stable cell line expressing the differential antigen was made and extensively characterized using a variety of biochemical and immunological assays. Preliminary data suggest prototype kits will not work with the alternate recombinant nonstructural antigen; however, a different diagnostic serology platform was piloted which could prove useful in an outbreak.
Successful development of new broadly available diagnostic tests for JEV provides the swine industry with the necessary tool to detect recent JEV exposure and/or infection in pigs, aiding biosecurity and control, which will help mitigate the spread and impact of a JEV outbreak in the US. As part of SHIC’s role in surveillance and discovery of emerging diseases, advancing diagnostic capabilities to detect JEV antibodies in swine helps to enhance preparedness for the US swine herd.
Foundation for Food & Agriculture Research
The Foundation for Food & Agriculture Research (FFAR) builds public-private partnerships to fund bold research addressing big food and agriculture challenges. FFAR was established in the 2014 Farm Bill to increase public agriculture research investments, fill knowledge gaps and complement the U.S. Department Agriculture’s research agenda. FFAR’s model matches federal funding from Congress with private funding, delivering a powerful return on taxpayer investment. Through collaboration and partnerships, FFAR advances actionable science benefiting farmers, consumers and the environment.
Swine Health Information Center
The Swine Health Information Center, launched in 2015 with Pork Checkoff funding, protects and enhances the health of the US swine herd by minimizing the impact of emerging disease threats through preparedness, coordinated communications, global disease monitoring, analysis of swine health data, and targeted research investments. As a conduit of information and research, SHIC encourages sharing of its publications and research. Forward, reprint, and quote SHIC material freely. For more information, visit http://www.swinehealth.org or contact Dr. Megan Niederwerder at [email protected] or Dr. Lisa Becton at [email protected].

When New World screwworm was first detected in Mexico in November 2024, USDA increased monitoring, prevention, and mitigation practices to address this emerging disease threat to domestic livestock. Those efforts include ongoing aerial dispersion of sterile NWS flies in Mexico and along the Texas border to curb fly population growth as well as active surveillance for NWS flies in border states. Monitoring swine cases in Mexico is an important component to evaluating risk to the US swine herd. As of April 1, 2026, over 1,270 swine have been affected with NWS in Mexico, representing approximately 6.5% of total cases.
NWS is a parasitic fly whose larvae feed on the living tissue of warm-blooded animals including livestock, pets, wildlife, and people. Larval infestations (myiasis) can occur in any broken or damaged skin and cause rapidly progressing, painful wounds that can lead to serious injury or death. Although NWS was eradicated from the US in the 1960s, the increasing number of confirmed cases in Central America and Mexico starting in 2024 has renewed concern about risks to swine health, human safety, and farm profitability.
In Texas alone, projections estimate that an NWS outbreak could result in more than $1.8 billion in livestock losses (USDA APHIS, 2025). Due to significant animal health and economic impacts, suspected cases of NWS must be reported to state animal health officials and the USDA immediately.
In total, reports from Mexico as of April 5, 2026, indicate that 19,677 head of livestock have been affected with 1,290 active cases, 120 of which were swine. Other active cases include 726 bovine, 309 canine, 72 equine, 37 sheep, 3 humans, 12 goats, six feline, and one bird. Accumulated totals of affected species since November 2024 include 13,217 bovine, 3,220 canine, 1287 swine, 962 equine, 517 sheep, 196 humans, 149 goats, 86 feline, and 30 birds.
NWS has not been found in the US since the 2016 – 2017 localized outbreak in Key deer in Florida. The concentration of NWS cases is in south and central Mexico; however, cases have been reported as closely as Nuevo Leon, just 70 miles south of the US border. The continued expansion of NWS case detections in Mexico highlights the critical need for continued vigilance to detect and report suspect cases in the US. The Swine Health Information Center supports producer awareness for this emerging disease through sharing regular NWS updates, hosting a webinar on the pest, and developing a new fact sheet and other resources for swine producers and other industry stakeholders.
Swine producers can prepare for NWS by understanding the fly’s behavior and life cycle, identifying production and housing conditions that increase the risk of myiasis, implementing effective biosecurity, environmental, and wound management practices, knowing the response procedures for suspected infestations, and developing an NWS disaster management plan.
On February 9, 2026, USDA announced the completion of a US-based NWS sterile fly dispersal facility in Edinburg, Texas. The announcement noted that the facility will expand USDA’s ability to disperse sterile flies along the border and into the US, if necessary. US Secretary of Agriculture Brooke Rollins stated, “This sterile fly dispersal facility was a high priority project, and our team delivered it in record time. This new facility is a monumental achievement for our domestic preparedness efforts, but we are also diligently working to stop the spread of screwworm in Mexico, conduct extensive trapping and surveillance along the border, increase US response capacity, and encourage innovative solutions. We will never stop fighting to protect American agriculture. USDA, through a whole-of-government approach, will continue to hold Mexico accountable to mitigating the spread of this dangerous pest.”
USDA has recently announced plans to start building a new sterile fly production facility in Edinburg, Texas later this year in addition to a $21 million investment to help renovate and convert an existing fruit fly facility in Metapa, Mexico. The Metapa, Mexico facility is expected to produce an additional 60 to 100 million sterile NWS flies per week. With continued support from USDA APHIS technical experts, Mexico expects production to begin there as early as summer 2026.
Providing publicly available, science-based information through fact sheets, monitoring reports, and stakeholder updates supports SHIC’s mission of minimizing the impact of emerging disease threats through coordinated communication of key resources and improving swine health information.
For more information, NWS resources are available here:

The Swine Health Information Center, in collaboration with the American Association of Swine Veterinarians, hosted a webinar entitled “Senecavirus A as an emerging disease risk for FMDV” on March 26, 2026 and is now available for on-demand viewing here. The webinar addressed Senecavirus A as a cause of vesicular disease in swine and the need for diligence to differentiate it from foot-and-mouth disease virus, a foreign animal disease of concern to US pork producers. The webinar had 221 registrants from 19 countries and was conducted in cooperation with the staff at the Swine Medicine Education Center at Iowa State University.
SVA was first identified in US swine in 2015 and is considered a significant health threat due to its similar clinical appearance to FMD. Recent monitoring has shown a change in SVA detection trends, warranting continued surveillance. Understanding the virus, its pathogenesis, routes of transmission, disease trends, as well as control and mitigation steps, can help producers protect their herd from this emerging disease and enhance prevention for other vesicular diseases such as FMD.
Key takeaways include:
These characteristics highlight the importance of surveillance, confirming diagnostics, and biosecurity measures to mitigate the impact of SVA and to differentiate it from high-consequence foreign animal diseases.
Presenters offering their expertise included:
Diego Diel, DVM, MS, PhD, Professor, Department of Population Medicine and Diagnostic Sciences, College of Veterinary Medicine, Cornell University
Ann Carpenter, DVM, MPH, DACVPM, Veterinary Medical Officer, Swine Health Team, Aquaculture, Swine, Equine, and Poultry Health Center, USDA APHIS Veterinary Services
Mariana Kikuti, DVM, MPH, PhD, Assistant Professor, Department of Veterinary Population Medicine, University of Minnesota
Rafael Romero Nicolino, PhD, Visiting Scholar, Iowa State University, Federal University of Minas Gerais, School of Veterinary Medicine – Brazil
Senecavirus A is a vesicular disease of swine belonging to the genus Senecavirus within the Picornaviridae family and has a small, positive-sense RNA genome of approximately 7.2 kb. SVA was first identified in 2002 as a contaminant in cell culture during investigations into cancer therapies. The emergence of SVA as a significant swine pathogen began in earnest around 2014, when outbreaks in Brazil caused vesicular disease clinically indistinguishable from FMD. The virus was then detected in the US mid-2015 with more than 5,000 confirmed cases documented since that time. This sharp increase underscores the growing importance of SVA in swine health and its role in triggering foreign animal disease (FAD) investigations due to its resemblance to FMD lesions.
Dr. Diel provided an overview for the clinical presentation of SVA noting that infection produces vesicular lesions on the snout, feet, and coronary bands of pigs, making it indistinguishable from other vesicular diseases like FMD without laboratory testing. This diagnostic challenge has major implications for regulatory responses, as any suspected FMD case requires mandatory reporting and investigation. Thus, detection of vesicular lesions from SVA contributes significantly to the burden of FAD investigations in the US swine industry.
Dr. Diel reviewed SVA pathogenesis and shared insights into the potential persistence in certain tissues and its role for disease transmission. Following inoculation, pigs develop a short-lived viremia typically lasting from approximately three to 10 days post-infection, with virus found in oral, nasal, and fecal secretions. Peak viral loads and shedding are observed early in infection, generally within the first week. SVA can be detected in multiple organs, but there is a notable tropism for lymphoid tissues, especially the tonsils. Virus can persist in tonsillar tissue beyond the acute infection phase and can be detected in animals for extended time periods. This finding is critical for understanding how SVA may persist within herds even after clinical signs have resolved.
Persistently infected animals can transmit SVA to naïve contacts even when low-level or subclinical infections occur and can contribute to disease spread within a population. In addition, SVA has been found in boar semen and highlights potential for semen-mediated transmission, particularly in systems using artificial insemination, which emphasizes the need for monitoring and biosecurity in boar studs.
Dr. Carpenter reviewed the challenges with differentiating SVA from other vesicular diseases of swine such as FMD. Mandatory reporting to USDA or a state animal health official is required if vesicular lesions are noted in swine. FAD investigations reflect seasonality of case detections with peaks occurring during summer months. Many of the investigations are overrepresented at slaughter establishments versus on-farm and cull sows represent a higher rate of investigation than market hogs. Standardized investigations are ongoing to determine the risk factors for disease detection and transmission in order to prevent further case presentation.
Dr. Carpenter provided an overview on the global FMD status and how the changing picture of disease detection across the globe is concerning. Various serotypes are identified in differing regions and the understanding of why a serotype departs from its existing “pool” of virus is limited. Vigilance for detection and diagnostic confirmation of vesicular lesions of swine continues to be critical to prevent FMD entry into the US especially with increasing global disease activity.
Dr. Kikuti reviewed the 10-year SVA trends for breeding herds from the SHIC-funded Morrison Swine Health Monitoring Project (MSHMP) data. Initially, a 2019 survey showed 17% prevalence of SVA in breeding herds and 7% in grow-finish. However, little data was available to determine how much virus circulation occurred in the US. The MSHMP data gathers case information from four diagnostic laboratories. Despite its clinical significance, the cumulative incidence of SVA in US breeding herds remained low, generally less than 2.5% per year across the 10-year study period. This suggests that while SVA continues to circulate, it affects a relatively small proportion of breeding herds annually. For sites experiencing more than one SVA outbreak, the median time interval between outbreaks was approximately 402 days, highlighting the potential for re-introductions and/or persistent circulation within herds.
Dr. Kikuti noted that a temporal pattern was observed, with peak SVA incidence occurring during the third and fourth quarters of the calendar year (July to December), suggesting seasonality influences disease transmission dynamics. A compilation of reports from multiple VDLs further supports this observation, with a consistent increase in the frequency of SVA cases during summer months. This seasonality requires further investigation and underscores the ongoing need for robust and collaborative surveillance systems that integrate on-farm observations with laboratory diagnostics to provide a comprehensive picture of SVA dynamics. Specifically, the observed seasonality and regional concentration suggest opportunities for more targeted biosecurity enhancements and surveillance efforts, particularly in the Midwest during the latter half of the year.
Dr. Nicolino provided an overview of Brazilian pork production, noting that farms are concentrated within the south, southeast and central regions of the country. SVA impacts swine of all ages with varying rates of morbidity and mortality depending upon the age of the pig. Three waves of SVA outbreaks occurred between 2014 to 2020 with differing regions affected. Piglets were impacted by SVA during the second wave of outbreaks in late 2018 with more than 50% of piglets exhibiting diarrhea. There is currently one vaccine commercially available for use in swine that has shown to increase antibodies 7-14 days after vaccination, but there has been limited evaluation of the effects post-challenge.
In Brazil, SVA surveillance is based on syndromic surveillance of disease, with all suspect cases being notifiable to animal health authorities. SVA in Brazil’s pork production follows a similar seasonal pattern to the US, reflective of warmer weather months. Risk factors contributing to this pattern are unknown but potential risks associated with infection could include animal movements, high replacement rates, poor sanitation and vector control. Dr. Nicolino noted that there is no current control program for SVA. Instead, producers focus on implementation of appropriate steps for FMDV prevention and utilize multiple biosecurity steps for those efforts.
More information on SVA research and resources can be found here.

In alignment with SHIC’s efforts to identify global swine disease threats to the US pork industry, the recent emergence and spread of FMDV SAT1 serotype is being closely monitored. The SHIC-funded Global Swine Disease Monitoring Reports, led by Dr. Sol Perez at the University of Minnesota, have highlighted the newly affected countries in monthly publications. To summarize the situation, Dr. Perez and her team have prepared the following article on the historic prevalence and current expansion of FMDV serotype SAT1, one of seven FMDV serotypes.
For FMDV, immunity is serotype-specific, meaning infection or vaccination against a given serotype does not confer protection against a different serotype. Historically, SAT1 has been confined to sub-Saharan Africa, with transmission largely maintained in endemic locations in East and Southern Africa. In 2025, SAT1 demonstrated a notable and concerning expansion beyond its traditional geographic range, with confirmed detections of two cocirculating subtypes (topotypes) of FMDV, SAT1/I and SAT1/III, across Western Asia and North Africa. The increasing circulation of FMDV serotype SAT1 poses a growing risk to previously unaffected regions, including South-East Europe and potentially beyond. As this serotype expands its geographic range, it creates additional pathways for introduction into new regions and countries, thereby increasing the overall likelihood of transboundary spread.
This briefing provides an overview of the historical range of the SAT1 serotype and analyzes its recent spread beyond established endemic zones. It also examines potential factors contributing to this dispersal and the resulting consequences for both endemic regions and countries free from FMD, such as the United States.
Historical distribution and epidemiologic context
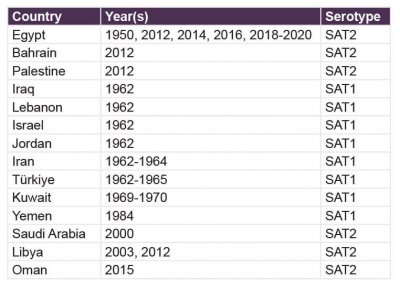
The historical occurrence of FMDV SAT serotypes outside sub-Saharan Africa has been relatively limited and characterized by sporadic incursions rather than sustained transmission. Early detections of SAT1 were reported across several countries in West Asia during the 1960s, including Iraq, Lebanon, Israel, Jordan, Iran, Türkiye, and Kuwait, followed by a later occurrence in Yemen in 1984. In contrast, SAT2 has been detected intermittently in North Africa and the Middle East, with notable events in Egypt, Bahrain, Palestine, Saudi Arabia, Libya, and Oman between 1950 and 2015. These historical patterns indicate that, before 2023, SAT serotypes were occasionally introduced into regions outside their endemic range but did not establish widespread or persistent circulation, in contrast to the more recent expansion observed in the current epidemiological landscape. The historical occurrence of FMD serotypes SAT1 and SAT2 is presented in table 1.
Table 1. Historical occurrence of FMDV SAT Serotypes outside sub-Saharan Africa (pre-2023)
Recent emergence and spread of FMDV SAT serotypes outside sub-Saharan Africa (2023-2026)
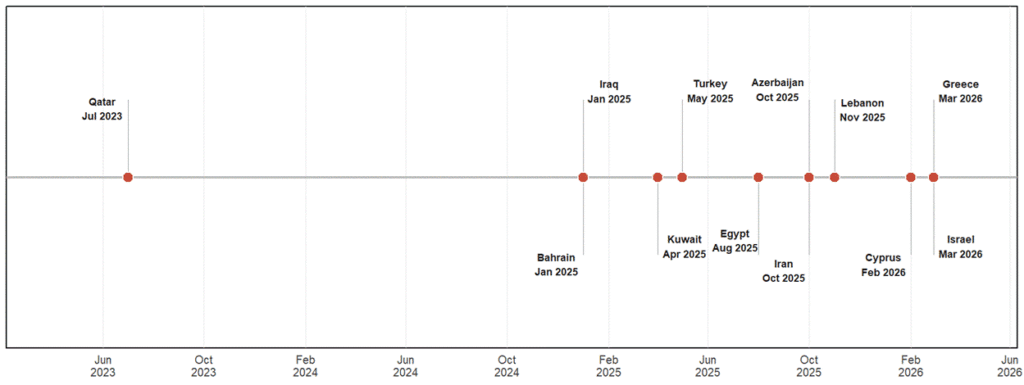
In 2023, confirmed reports of FMDV serotype SAT1 were reported in Qatar and Jordan, and SAT2 in Oman, Türkiye, and Bahrain. From 2025 onward, SAT1 expanded rapidly, with confirmed detections in Iraq, Kuwait, Türkiye, and Egypt, followed by further spread into Azerbaijan, indicating northward expansion into the Caucasus. Continued transmission was observed with subsequent detections in Lebanon, Israel, Cyprus, and Greece in 2025-2026. During this period, SAT2 persisted in Algeria and Bahrain, reflecting ongoing co-circulation in some regions. These patterns suggest a transition from sporadic SAT detections to a broader and more sustained geographic presence of SAT1 across the Eastern Mediterranean, West Asia, and North Africa. Figure 1 presents a timeline of SAT1 outbreaks occurring outside endemic regions between 2023 and 2026, while Figure 2 is a map illustrating the countries affected by both SAT1 and SAT2 during that same timeframe.
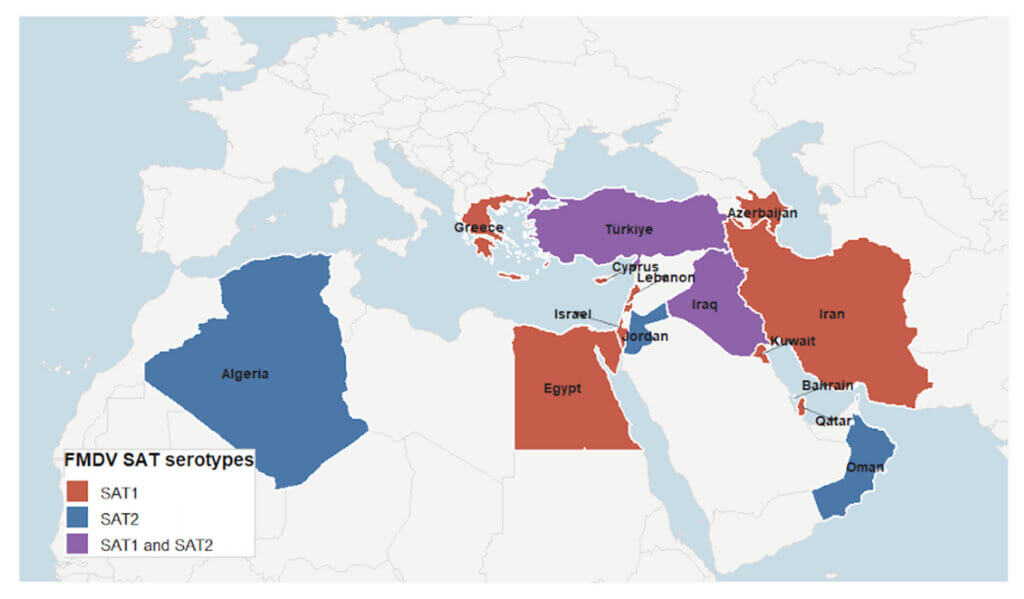
Implications for disease control in the affected countries
The emergence of FMDV serotype SAT1 across the eastern Mediterranean, West Asia, and North Africa exposes critical vulnerabilities in existing control systems within endemic countries. Most vaccination programs in these regions are designed around historically occurring serotypes O, A, and Asia-1, providing no cross-protection against SAT1 and leaving livestock populations effectively susceptible despite routine vaccination. This risk is compounded by the limited availability of SAT1-containing vaccines, gaps in surveillance, and variable veterinary service capacity. A recent FAO rapid risk assessment further highlights that informal cross-border animal movements, particularly of small ruminants that may be subclinically infected, are a major driver of transmission, with seasonal increases in livestock movement during religious periods further amplifying spread. Together, these factors create conditions for rapid and often undetected dissemination of SAT1, which increases the likelihood of unexpected outbreaks, reduces the effectiveness of current control strategies, and presents a significant economic and food security risk, particularly in smallholder-dominated systems.
Potential drivers of SAT1 spread
The expansion of FMDV serotype SAT1 is likely due to several interconnected factors. Increased informal transboundary livestock movement is a primary driver of rapid dissemination. Environmental pressures, such as drought and land-use change, enhance spillover opportunities from wildlife reservoirs into domestic herds at the wildlife-livestock interface. Gaps in vaccination strategies, which often target serotypes O and A, leave populations vulnerable and create ecological space for SAT1 to spread. Although improved surveillance contributes to detection, the speed and extent of the recent spread point to a true epidemiological expansion, not merely an artefact of reporting.
Implications for the United States
The ongoing expansion of FMDV serotype SAT1 has important implications for FMD-free countries, including the United States, because it increases both the complexity and uncertainty of global FMD risk. The emergence of SAT1 in new regions adds to the diversity of circulating serotypes in those regions, complicating preparedness and response planning, particularly when vaccine stockpiles may not include well-matched SAT1 strains. At the same time, continued transboundary spread heightens the risk of introduction through established pathways such as international travel, contaminated animal products, and fomites. Although the United States remains free of FMD, these global developments underscore the need to strengthen early detection and surveillance systems, maintain stringent biosecurity measures across livestock value chains, and ensure that vaccine preparedness strategies are sufficiently flexible to incorporate emerging serotypes such as SAT1.
References

The Swine Health Information Center partnered with the Foundation for Food & Agriculture Research and the Pork Checkoff to fund a $4 million research program to enhance prevention, preparedness, mitigation, and response capabilities for H5N1 influenza in the US swine herd. The first RFP was announced in November 2024 and resulted in 10 H5N1 research projects being funded in 2025 for a total of $2.2 million. A second RFP was announced in January 2026 outlining seven priorities not yet adequately addressed in the first 10 projects. By March 2026, a total of 42 proposals were received from 26 different organizations across three countries. Proposals will undergo competitive review for funding recommendations based on value to US pork producers.
The continued outbreaks of H5N1 influenza in livestock, poultry and other species highlight the potential threat of this pathogen to the US swine industry. Research priorities for the second H5N1 Risk to Swine RFP are designed to further strengthen US swine industry prevention and preparedness as well as inform response efforts should H5N1 be introduced into the commercial swine herd. The 42 proposals received are expected to address H5N1 Risk to Swine research priorities described in the detailed Request for Research Proposals including topic areas of 1) diagnostic surveillance, 2) introduction and transmission risks, 3) caretakers of pigs, 4) biosecurity practices, 5) safety of pork, 6) mitigating production impact, and 7) business continuity.
Upon completion of the competitive review process, project awards are expected to be announced in the summer of 2026. Projects demonstrating the most urgent needs and timeliness of completion, providing the greatest value to US pork producers, and showing efficient use of funds will be prioritized for funding. Results will be shared with producers and veterinarians as soon as they become available.
Critical research investments are necessary to understand and prevent H5N1 incursion, ensure rapid detection of H5N1 if introduced, protect animal and caretaker health, inform stakeholder response, mitigate production losses on the farm, identify effective control measures, and develop clear messaging to consumers on the safety of pork. Outcomes from the funded proposals will provide critical information that producers, veterinarians, and industry stakeholders can use to better prevent incursion and develop preparedness plans if H5N1 is identified in commercial swine herds within the US.
Foundation for Food & Agriculture Research
The Foundation for Food & Agriculture Research (FFAR) builds public-private partnerships to fund bold research addressing big food and agriculture challenges. FFAR was established in the 2014 Farm Bill to increase public agriculture research investments, fill knowledge gaps and complement the U.S. Department Agriculture’s research agenda. FFAR’s model matches federal funding from Congress with private funding, delivering a powerful return on taxpayer investment. Through collaboration and partnerships, FFAR advances actionable science benefiting farmers, consumers and the environment.
Swine Health Information Center
The Swine Health Information Center, launched in 2015 with Pork Checkoff funding, protects and enhances the health of the US swine herd by minimizing the impact of emerging disease threats through preparedness, coordinated communications, global disease monitoring, analysis of swine health data, and targeted research investments. As a conduit of information and research, SHIC encourages sharing of its publications and research. Forward, reprint, and quote SHIC material freely. For more information, visit http://www.swinehealth.org or contact Dr. Megan Niederwerder at [email protected] or Dr. Lisa Becton at [email protected].

This month’s Domestic Swine Disease Monitoring Report highlights a slight decrease in PEDV case positivity in wean-to-market from February. However, adult/sow farm PEDV case positivity has remained on an upward trend since October 2025. PDCoV case positivity has also continued to increase since late 2025. Notably, PRRSV case positivity in the wean-to-market category has been trending downward since November 2025; however, it reached 44.6% in the first quarter of 2026, marking the highest first-quarter level observed over the past decade. This month’s bonus page highlights insights from two decades of PRRSV evolution. The accompanying podcast features Dr. Deb Murray, director of veterinary services at New Fashion Pork, who discusses the increasing severity and rapid spread of PRRSV driven by highly virulent emerging variants, along with a resurgence of PED and its impact on pig mortality across production stages. She explains that while full disease elimination remains challenging, applying a comprehensive package of biosecurity, vaccination, and coordinated management practices is key to reducing transmission and production losses.

In this month’s Global Swine Disease Monitoring Report, read about foot-and-mouth disease serotype SAT1 being reported for the first time in Gansu Province and the Xinjiang Uyghur Autonomous Region in China. In Greece, the first FMD outbreak in 25 years was confirmed on Lesvos. Rapid expansion occurred with over 17 outbreaks confirmed in Greece as of March 27. The first case of African swine fever was confirmed in wild boar in Hochsauerland district, North Rhine-Westphalia, Germany. In South Korea, the government intends to strengthen ASF response after the virus was detected in livestock feed. A special edition showcasing the 100th Global Swine Disease Monitoring Report highlights the evolution of the surveillance report since 2017 and invites stakeholder feedback.
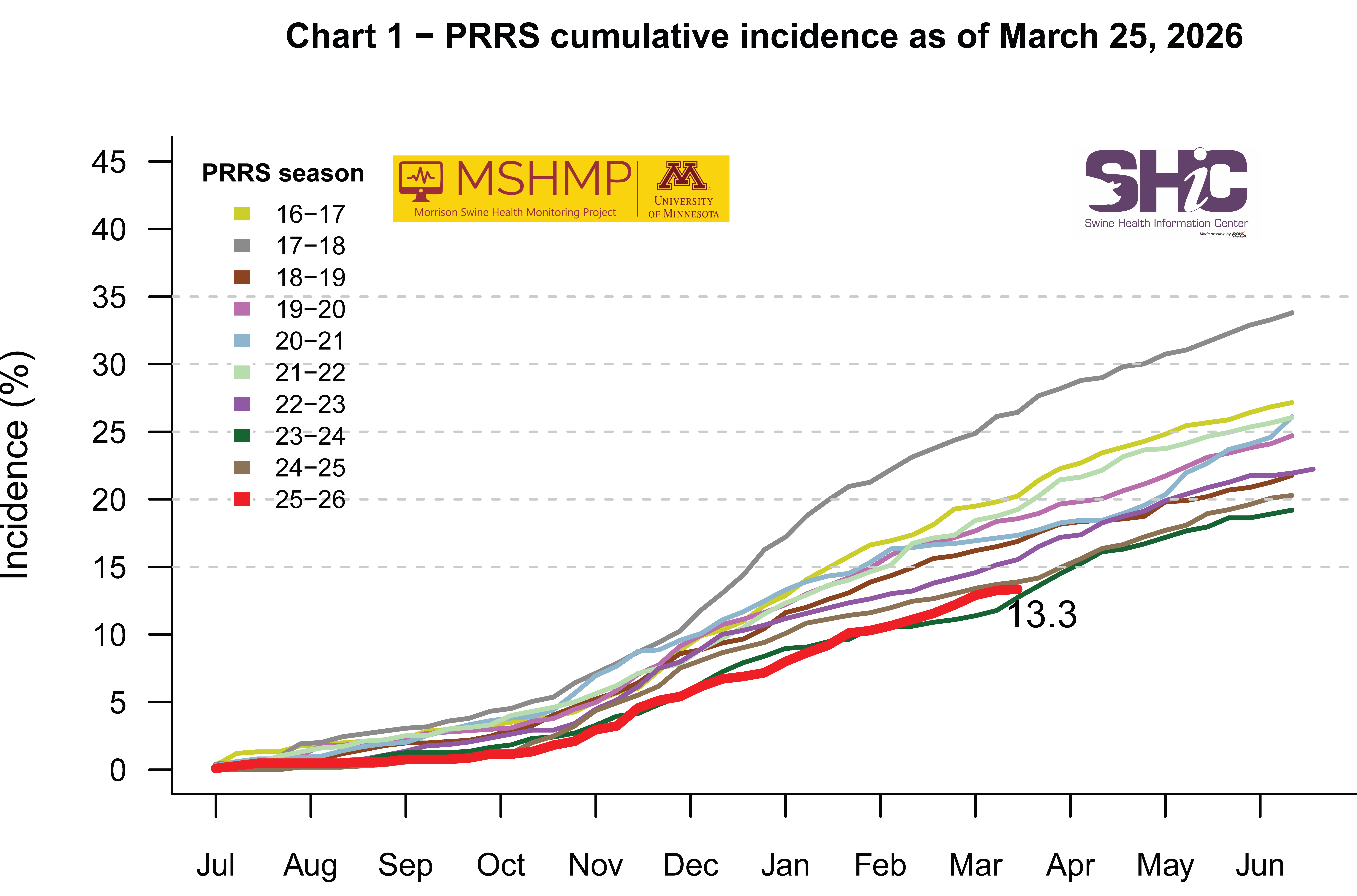
PRRS Cumulative Incidence for MSHMP
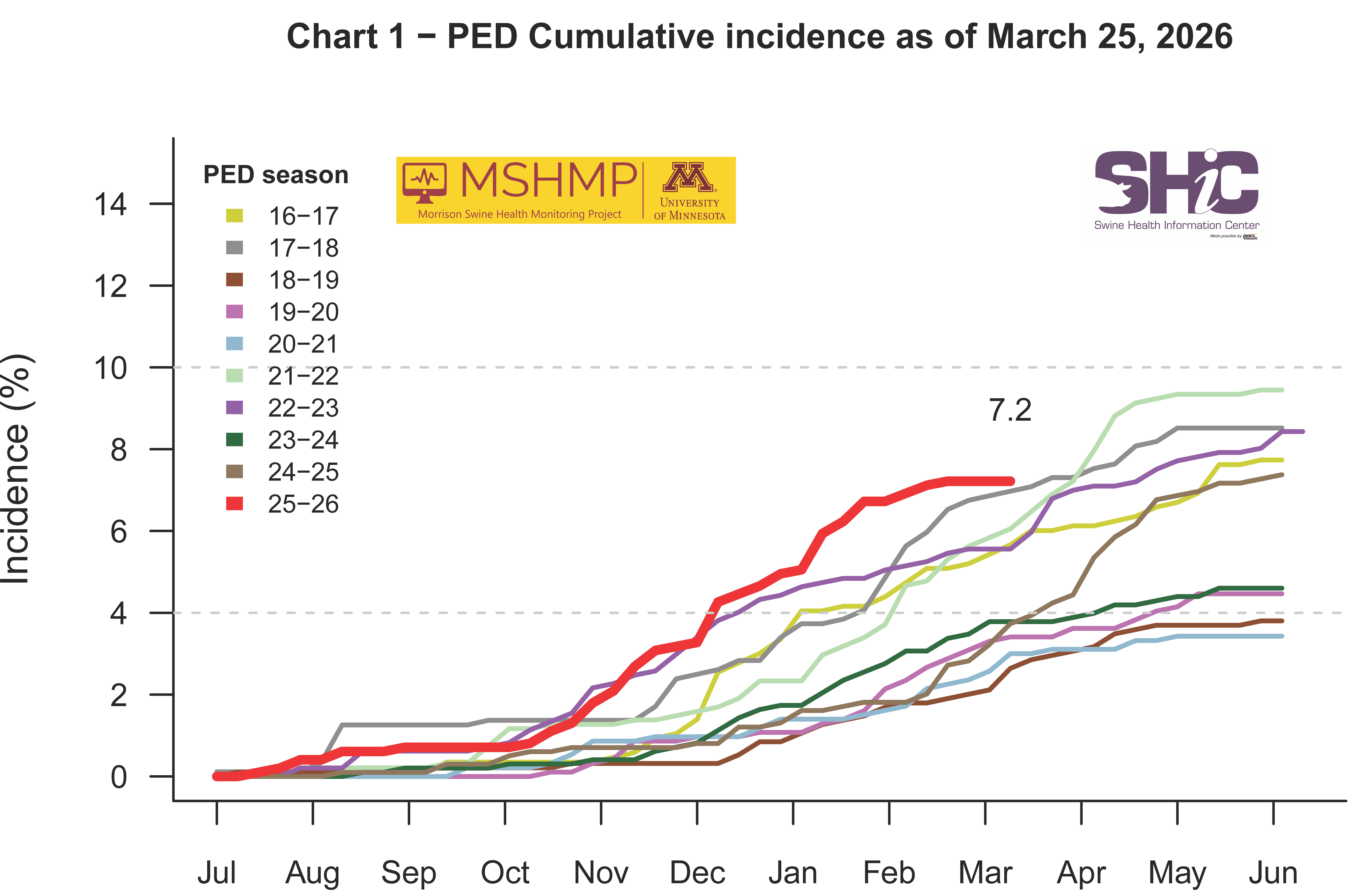
PEDV Cumulative Incidence for MSHMP

Five PRRSV variants are classified as Variants Under Monitoring (VUM) Category 2 or higher in this month’s report. Variants 1C.5 and 1C.5.35 were promoted to VUM Categories 4 and 3, respectively. Variants 1C.5.32 and 1H.18 remain at VUM Categories 4 and 2, respectively, while Variant 1C.5.33 was demoted to VUM Category 1. These changes are reflected in new variant-specific situation reports). Previous reports for all variants ever classified as VUM Category 2 or higher remain available.